Partnership agreement was made in 2002 to form a software engineering team in the field of digital medical imaging, archiving and organization of radiology wards.
Company was founded in 2003 and registered in Genex tower, Belgrade Serbia on the 27th floor.
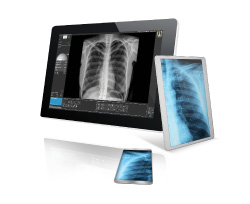
The first product to be developed was Visaris PACS and Diagon and first installation done in South Africa. Development of Visaris RIS began immediately after that in early 2004.
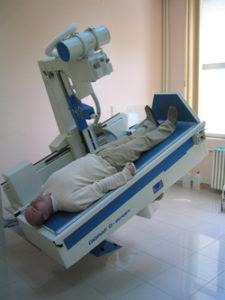
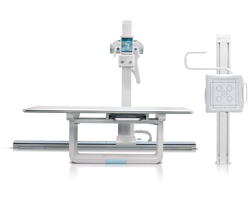
In the same year, Trixell’s flat panel detector was bought and integrated to construct the first Serbian digital X ray machine Digraf X installed in an orthopaedics clinic.